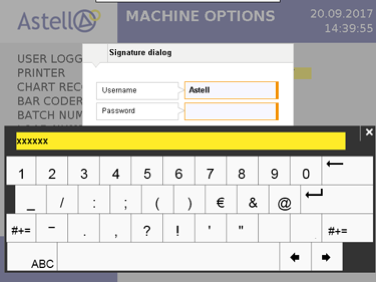
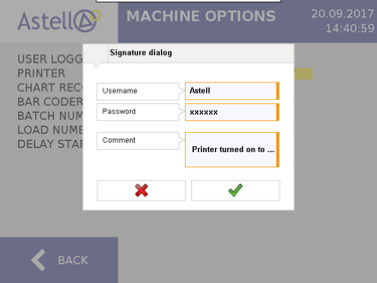
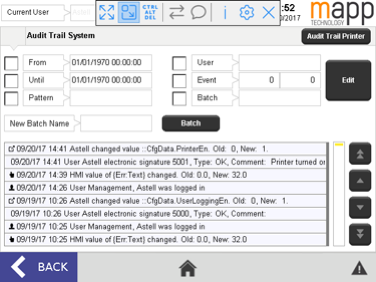
Astell is able to provide EDS controller software that conforms to FDA 21 CFR Part 11 guidelines. The fundamental requirements of 21 CFR 11 are the compliance with Data Security, Data Integrity, Traceability/Audit Trails and Electronic Signature rules. In the case of sterilizers, this means any change made to a cycle, option, or device parameter will require an electronic signature. All device changes and signatures will be electronically logged and can be recalled to provide a clear picture of user input for any given point in time. All audit trails are saved in a file format that cannot be altered in any way.
To achieve this level of compliance requires a dynamic and robust controller, such as the colour touchscreen system Astell provides on its Effluent Decontamination Systems. By incorporating all device settings in to one electronic interface users have the necessary control and security at their fingertips.